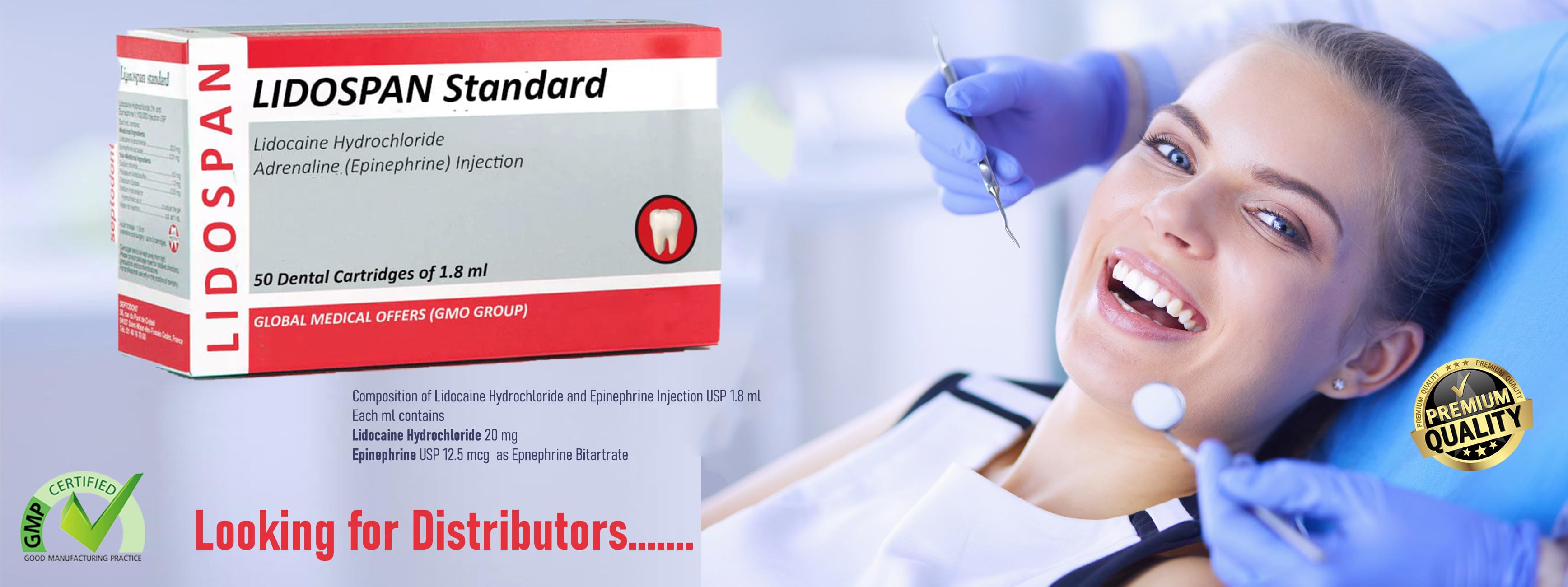
LIDOSPAN® Dental Cartridge — Reliable Analgesia, Proven Safety
LIDOSPAN® Dental Cartridge — Reliable Analgesia, Proven Safety
Manufactured in India • Marketing Authorization Holder: GLOBAL MEDICAL OFFERS
Effective, predictable local anesthesia is the backbone of modern dental practice. LIDOSPAN®, a sterile cartridge formulation of 2% lidocaine hydrochloride with epinephrine, is designed to deliver fast onset, dependable pulpal and soft-tissue anesthesia, and an established safety profile — now marketed by GLOBAL MEDICAL OFFERS. Below is a concise, evidence-based summary clinicians and procurement teams can use when evaluating LIDOSPAN for routine and operative dentistry.
Pharmacology & clinical rationale
Lidocaine (2% solution) is an amide local anesthetic with a rapid onset and intermediate duration of action. The addition of epinephrine (commonly 1:100,000 or 1:50,000) provides vasoconstriction that prolongs local effect, reduces systemic absorption, and improves hemostasis for surgical procedures. Lidocaine remains one of the most widely used and studied dental local anesthetics worldwide. DailyMedDrugBank
Onset and duration — what to expect with LIDOSPAN
-
Onset of anesthesia: For dental nerve blocks and infiltrations, lidocaine with epinephrine typically shows clinical onset in about 2–4 minutes, allowing clinicians to begin procedures quickly. FDA Accessdatadarbydental.com
-
Pulpal anesthesia duration: Studies and product monographs report pulpal anesthesia commonly lasting ~60 to 90 minutes after standard dental administration (exact time depends on injection type — infiltration vs nerve block — and epinephrine concentration). Some sources report pulpal anesthesia decline starting around 49–53 minutes in certain infiltration scenarios, while nerve blocks often provide longer pulpal coverage. PMCdarbydental.com
-
Soft tissue anesthesia: Soft tissue numbness frequently persists ~2 to 2.5 hours, giving clinicians postoperative analgesic coverage for minor procedures. FDA Accessdata
Pharmacokinetics — half-life and systemic profile
Lidocaine’s elimination after parenteral administration is characteristically intermediate, with the elimination half-life most commonly cited at ~1.5–2.0 hours in healthy adults. Lidocaine is extensively metabolized in the liver to active and inactive metabolites; impaired hepatic function prolongs elimination and requires dose adjustments. FDA AccessdataPubMed
Pregnancy & special populations
-
Pregnancy category: Lidocaine is generally classified as FDA Pregnancy Category B in injectable formulations; animal studies have not demonstrated teratogenicity at therapeutic exposures, but clinicians should use the lowest effective dose and exercise clinical judgement. National dental guidance supports lidocaine (with appropriate precautions) as a preferred agent during pregnancy when local anesthesia is required. FDA AccessdataPMC
Efficacy — how lidocaine compares (brief context)
While lidocaine remains a mainstay for dental anesthesia, several meta-analyses show that 4% articaine can have higher rates of anesthetic success than 2% lidocaine for infiltration techniques. This does not negate lidocaine’s established role — it remains reliable for nerve blocks, infiltrations, and many everyday dental procedures — but it helps clinicians select the optimal agent depending on technique and clinical goal. PubMedPMC
Cartridge design & safety features (manufacturer-declared)
LIDOSPAN cartridges are manufactured with modern cartridge safety and usability in mind (manufacturer-declared features commonly requested by procurement and clinicians):
-
No-methylparaben formulation — avoids methylparaben preservatives that can cause hypersensitivity in susceptible patients. darbydental.com
-
Latex-free plungers and seals — reduces risk for latex-sensitive patients and staff (aligns with contemporary cartridge component standards). Septodont USA
-
Terminal sterilization & safe packaging — cartridges are terminally sterilized and packaged (e.g., blister trays, Mylar labels on glass cartridges) to minimise breakage and ensure sterility until use. Septodont USAdarbydental.com
For procurement or regulatory purposes, request LIDOSPAN’s Certificate of Analysis (CoA), product monograph, and batch release/sterilization certificates from GLOBAL MEDICAL OFFERS to verify these features for each lot.
Practical quick-reference (clinician’s snapshot)
-
Active ingredients: Lidocaine HCl 2% + Epinephrine (typical 1:100,000 or 1:50,000). DailyMed
-
Onset: ~2–4 minutes. FDA Accessdata
-
Pulpal anesthesia: ~60–90 minutes (varies by technique). PMC
-
Soft tissue anesthesia: ~2–2.5 hours. FDA Accessdata
-
Half-life: ~1.5–2.0 hours (may be prolonged in hepatic impairment). FDA Accessdata
-
Pregnancy: FDA Category B — use when clinically indicated with appropriate precautions. FDA Accessdata
Summary
LIDOSPAN® provides clinicians a time-tested, predictable option for dental local anesthesia: rapid onset, dependable duration for pulpal and soft-tissue procedures, and a well-characterized safety profile. Manufactured in India and distributed under Marketing Authorization by GLOBAL MEDICAL OFFERS, LIDOSPAN cartridges combine pharmacologic reliability with contemporary cartridge design (no-methylparaben, latex-free components, terminal sterilization) to support safe use across typical dental populations.
Selected certified references
-
Xylocaine® Dental (Lidocaine HCl) product labeling (FDA / AccessData). FDA Accessdata+1
-
Lidocaine Hydrochloride and Epinephrine, USP — DailyMed / product monograph. DailyMed
-
Pulpal anesthesia duration studies and clinical reports (PMC / peer-reviewed articles). PMC
-
Pharmacokinetics and elimination half-life data (FDA label; pharmacokinetic reviews). FDA AccessdataPubMed
-
Cartridge technical/packaging notes from dental suppliers (statements on terminal sterilization, Mylar labels, latex-free components). Septodont USAdarbydental.com