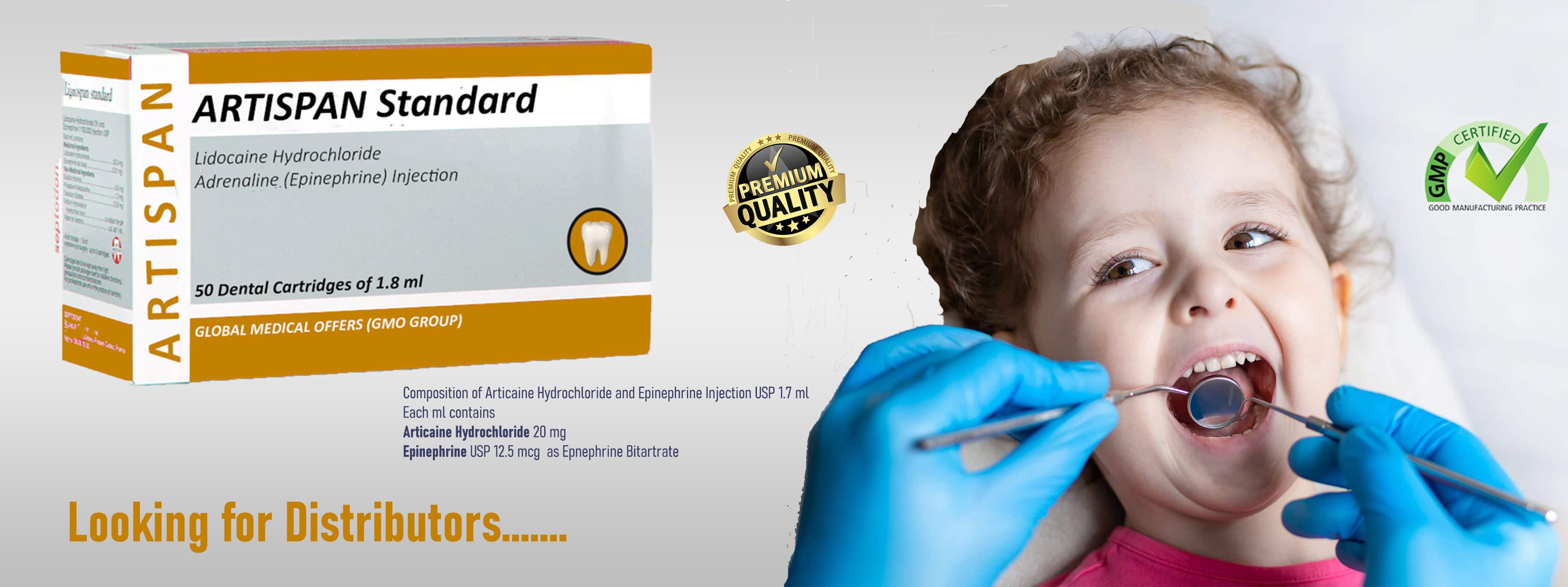
ARTISPAN® Dental Cartridge — Precision Analgesia, Trusted Safety
ARTISPAN® Dental Cartridge — Precision Analgesia, Trusted Safety
Manufactured in India • Marketing Authorization Holder: GLOBAL MEDICAL OFFERS
Pain control in dentistry demands local anesthetics that act fast, last predictably, and carry an excellent safety profile. ARTISPAN® — a 4% articaine hydrochloride formulation with epinephrine (vasoconstrictor) — was developed precisely to meet those needs in modern dental practice. Manufactured in India and marketed by GLOBAL MEDICAL OFFERS, ARTISPAN combines proven clinical performance with cartridge design choices that reduce allergen exposure and improve procedural safety.
Why articaine + epinephrine?
Articaine is an amide local anesthetic widely used in dentistry because of its unique pharmacokinetic and clinical advantages: faster onset, strong tissue penetration, and a comparatively short metabolic half-life. When combined with epinephrine (commonly 1:100,000 or 1:200,000), clinicians obtain more reliable anesthesia and improved hemostasis for surgery and operative procedures. Clinical meta-analyses show articaine provides higher anesthetic success than lidocaine for many infiltration and supplemental techniques — an evidence base that supports its contemporary use. NaturePubMed
Fast onset — minimise waiting, speed treatment
ARTISPAN’s articaine/epinephrine formulation produces clinically meaningful anesthesia quickly: onset is typically within 1–9 minutes after injection, helping to avoid delays before effective analgesia. Infiltrations generally become effective within that window, allowing procedures to start sooner with greater patient comfort. Pfizer Labeling
Predictable duration — plan your procedure with confidence
Clinical and prescribing information report typical durations as follows:
-
Pulpal anesthesia: around ~60 minutes for infiltrations; longer (up to ~120 minutes) for nerve blocks in many patients.
-
Soft tissue anesthesia: often lasts considerably longer (commonly around 1–2 hours), giving postoperative coverage for minor discomfort. These values help clinicians select ARTISPAN for both routine and more complex interventions. Pfizer LabelingDailyMed
Pharmacokinetics — rapid elimination, favorable profile
Articaine is notable for a short metabolic half-life — often reported in the literature at approximately 27–42 minutes — which is shorter than many other amide anesthetics. A shorter half-life can reduce systemic exposure while preserving effective local block, an important consideration when treating medically complex patients. PMC
Efficacy — clinically meaningful advantages
Multiple randomized trials and systematic reviews indicate that 4% articaine (with epinephrine) is more likely than 2% lidocaine to achieve successful anesthesia for infiltration techniques and is frequently used as an effective supplemental infiltration after a failed inferior alveolar nerve block. Reported anesthetic success rates for 4% articaine range broadly in clinical contexts (e.g., ~61–83% in some endodontic settings), and pooled analyses report significantly higher odds of success versus lidocaine for infiltrations (several-fold greater likelihood in meta-analyses). These data explain why many practitioners prefer articaine for both maxillary and mandibular infiltrations and for supplemental techniques. peerj.comAmerican Association of Endodontists
Safety & special populations
-
Adverse effects: Large systematic reviews and pediatric analyses show comparable rates of adverse events for articaine versus other commonly used local anesthetics, when used appropriately. Clinicians should always aspirate prior to injection and adhere to recommended dosing limits. Journal of Clinical Pediatric Dentistry
-
Pregnancy: Historically categorized as FDA Pregnancy Category C (recommendation: use only if clearly needed and after risk–benefit discussion). Modern practice emphasizes careful selection and the lowest effective dose in pregnancy; consult local guidelines. PMCDailyMed
ARTISPAN cartridge design — safety and allergen risk reduction
ARTISPAN cartridges are engineered for routine dental workflow and patient safety. The following product attributes are manufacturer-declared (and align with best practices used by leading dental cartridge manufacturers):
-
No-methylparaben formulation — avoids methylparaben preservatives that can be associated with hypersensitivity reactions in susceptible patients. (Manufacturer-declared; aligns with preservative-free/no-methylparaben formulations used by several established brands.)
-
Latex-free plungers and seals — to minimise the risk of latex allergy in sensitive patients and staff; many cartridge producers now use latex-free components.
-
Terminal sterilization of cartridges — cartridges undergo terminal sterilization processes to ensure product sterility and integrity before release
Practical summary for clinicians
-
Onset: typically 1–9 minutes — rapid clinical onset helps reduce chairtime. Pfizer Labeling
-
Pulpal anesthesia duration: infiltration ~60 min; nerve block up to ~120 min in many cases. Pfizer Labeling
-
Soft tissue anesthesia: commonly ~1–2 hours depending on technique and epinephrine concentration. Pfizer Labeling
-
Half-life: ~27–42 minutes (favourable for limiting systemic exposure). PMC
-
Pregnancy category / use: historically Category C — exercise clinical judgement and consult current guidance. PMCDailyMed
-
Clinical advantage: articaine shows higher likelihood of anesthetic success for infiltrations versus lidocaine in multiple meta-analyses (ORs ranging from ~2–4 depending on the technique and study set). NatureAmerican Association of Endodontists
About GLOBAL MEDICAL OFFERS (MAH)
GLOBAL MEDICAL OFFERS is the Marketing Authorization Holder for ARTISPAN. As MAH, the company is responsible for regulatory compliance, product information, pharmacovigilance reporting, and distribution oversight in the territories where ARTISPAN is authorized. For formal procurement, clinical documentation, product monographs, or batch certificates, request the ARTISPAN dossier and regulatory documentation directly from GLOBAL MEDICAL OFFERS.
References
-
Articaine HCl and Epinephrine — prescribing / product information (DailyMed / FDA label): onset and duration; dosages. DailyMedPfizer Labeling
-
Scholarly review: “The potential of articaine as new generation of local anesthesia” — pharmacokinetics and half-life discussion. PMC
-
Meta-analysis / systematic review — articaine vs lidocaine: pooled analyses showing higher anesthetic success for articaine in infiltrations. NatureAmerican Association of Endodontists
-
Product pages and technical notes from cartridge manufacturers and dental suppliers — statements on no-methylparaben, latex-free components, and terminal sterilization as contemporary cartridge features (examples from dental suppliers/manufacturers). pattersondental.comDentalkart
-
Guidance article on pregnancy considerations and local anesthetics in dentistry. PMC