"Buying ‘Miracle’ Ozempic Online Could Put Your Life at Risk
The frenzy surrounding semaglutide—a
breakthrough GLP‑1 “miracle” weight‑loss injection—is escalating, and the
danger of counterfeit versions is more apparent than ever.
1. The Shortage Has Ended… Mostly
As of February 21, 2025, the U.S.
Food and Drug Administration declared that the national shortage of
semaglutide (marketed as Ozempic and Wegovy) has been resolved,
meaning that manufacturer supply can now meet national demand Foley & Lardner LLPGoodRx.
Despite this, localized supply constraints
remain possible as products move through the supply chain U.S. Food and Drug AdministrationCollege of Public Health. Similarly, in Australia,
the Therapeutic Goods Administration confirms that prescribing restrictions
have been lifted and availability is improving Therapeutic Goods Administration (TGA).
2. Compounded ‘Copycat’ Versions Banned
During the shortage, compounding pharmacies
legally produced semaglutide replicas under FDA enforcement discretion. That
temporary allowance has now ended:
- 503A pharmacies: allowed until April 22, 2025
- 503B outsourcing
facilities: until May
22, 2025 Burr & FormanSELF
Post-deadline, only brand‑name, FDA‑approved
medications are legal, except for exceptional, patient‑specific
modifications SELFBurr & Forman.
3. Surge in Legal Action Against Unauthorized
Compounders
Novo Nordisk has ramped up litigation, filing 14
new lawsuits against compounding pharmacies, telehealth firms, and medical
spas accused of selling unauthorized semaglutide—often with illicit ingredients
and no proof of safety Reuters.
These lawsuits reflect growing concern over unsafe,
unverified knockoffs, which may result in serious health risks and legal
violations Investors.comReuters.
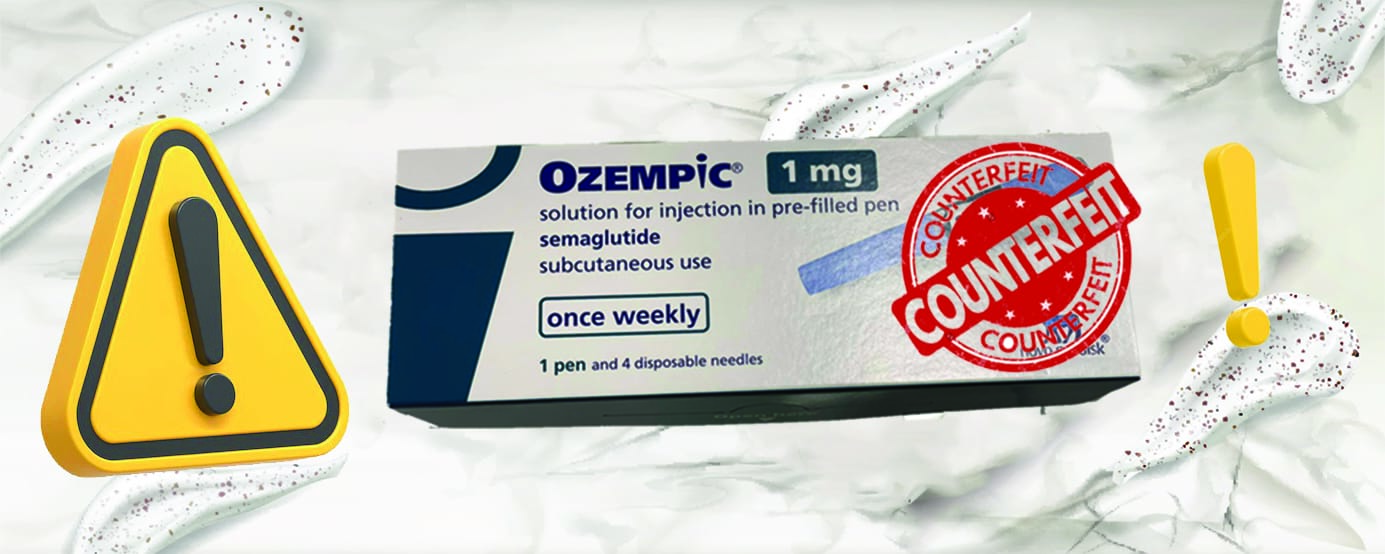
4. Counterfeit Ozempic Entering the Supply
Chain
In April 2025, the FDA and Novo Nordisk
warned that several hundred counterfeit units of Ozempic (semaglutide
1 mg) had entered the drug supply chain in the U.S. These were seized on April
9, but their identity, quality, and safety remain under investigation U.S. Food and Drug AdministrationNew York PostABC7 Los Angeles.
Products bearing lot number PAR0362 and
serial numbers starting with 51746517 are confirmed as counterfeit U.S. Food and Drug AdministrationNew York PostABC7 Los Angeles. While six adverse events have been linked
to that batch, none are yet conclusively tied to counterfeit drug use U.S. Food and Drug AdministrationNew York Post.
5. Global Concerns, Including the UK and Beyond
In the UK, the MHRA seized 400 pre‑filled
pens in 2024—quintupling the previous year's total—amid a rising black
market for semaglutide sold to ineligible patients via illicit online sources The Times.
The World Health Organization advises
that semaglutide should only be obtained through qualified healthcare
professionals diabetes.org.uk. MHRA and NHS guidance reiterates that GLP‑1
drugs are licensed for specific medical conditions—not as cosmetic or
convenience weight‑loss aids—and that misuse poses serious risks GOV.UKRegulatory Affairs.
Additionally, the UK is tightening controls on
online pharmacies prescribing weight‑loss jabs like Wegovy and Ozempic to
prevent misuse and unregulated sales The Guardian.
6. Affordable Generics on the Horizon—but with
Caution
India is preparing to release generic semaglutide by March 2026, which
could slash prices by up to 80%, a major shift in accessibility The Economic Times. Companies like Eris Lifesciences
are positioning to lead this generic wave The Economic Times.
In Canada, Sandoz aims to reduce
prices of semaglutide drugs by about 70% post-patent expiry in early
2026, despite concerns that U.S. demand may strain Canadian supply Financial Times.
Final Word: The Bottom Line
- The semaglutide shortage is
officially over—but compounded knockoffs remain banned.
- Counterfeit products are
still circulating, posing
serious health and safety risks.
- Regulatory agencies worldwide
are cracking down—but so are online black‑market sellers.
- Genuine, affordable
generics are on the way,
but until then, authorized medical supervision and pharmacy channels
are paramount.